Slide Preparation & Staining
Lesson 14 of 16 · Detailed theory + identification points
Points of Identification
4 pointsDetailed Theory
Object: Demonstration, Preparation and Staining of Histological Slides
Introduction
Histological slide preparation converts living (or fixed) tissue into a thin, transparent, stained section mounted on a glass slide suitable for light microscopy. The standard process consists of several sequential steps.
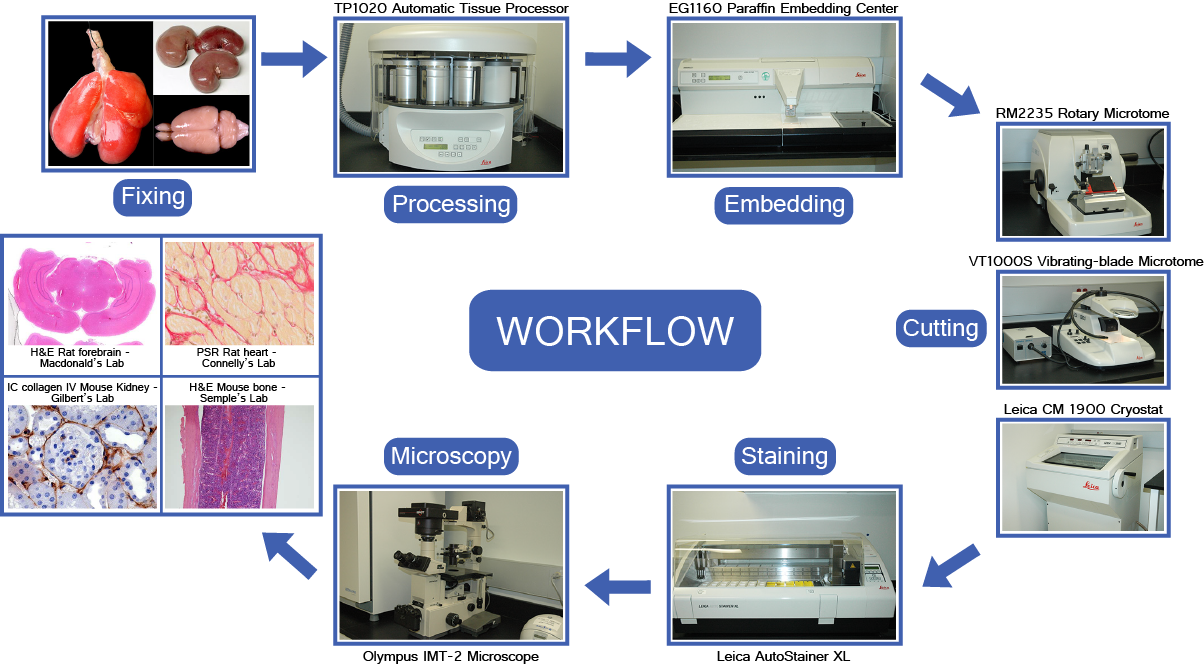
STEP 1 — Fixation
Purpose: preserve tissue architecture and cell morphology; prevent autolysis and putrefaction; harden tissue for sectioning.
- 10% Neutral Buffered Formalin (NBF): most widely used — cross-links proteins via methylene bridges; volume ratio of fixative to tissue = 10:1; duration 6–24 hours depending on tissue size
- Glutaraldehyde: superior cross-linker for electron microscopy (EM)
- Bouin's fluid: picric acid + formalin + acetic acid; excellent for testis and small GI biopsies; yellow colour
- Zenker's fluid: mercuric chloride-based; good nuclear detail; for haematological tissues
STEP 2 — Grossing and Tissue Processing
- Grossing: macroscopic examination; representative sections selected; placed in labelled cassettes
- Dehydration: ascending grades of ethanol (70% → 80% → 95% → 100% × 2) removes water from tissue
- Clearing: xylene (or xylene substitute) replaces alcohol; renders tissue transparent; allows paraffin infiltration
- Infiltration: tissue immersed in molten paraffin wax (56–60°C) under vacuum — wax replaces xylene
- Embedding: tissue oriented in mould with molten wax; cooled to produce a paraffin block
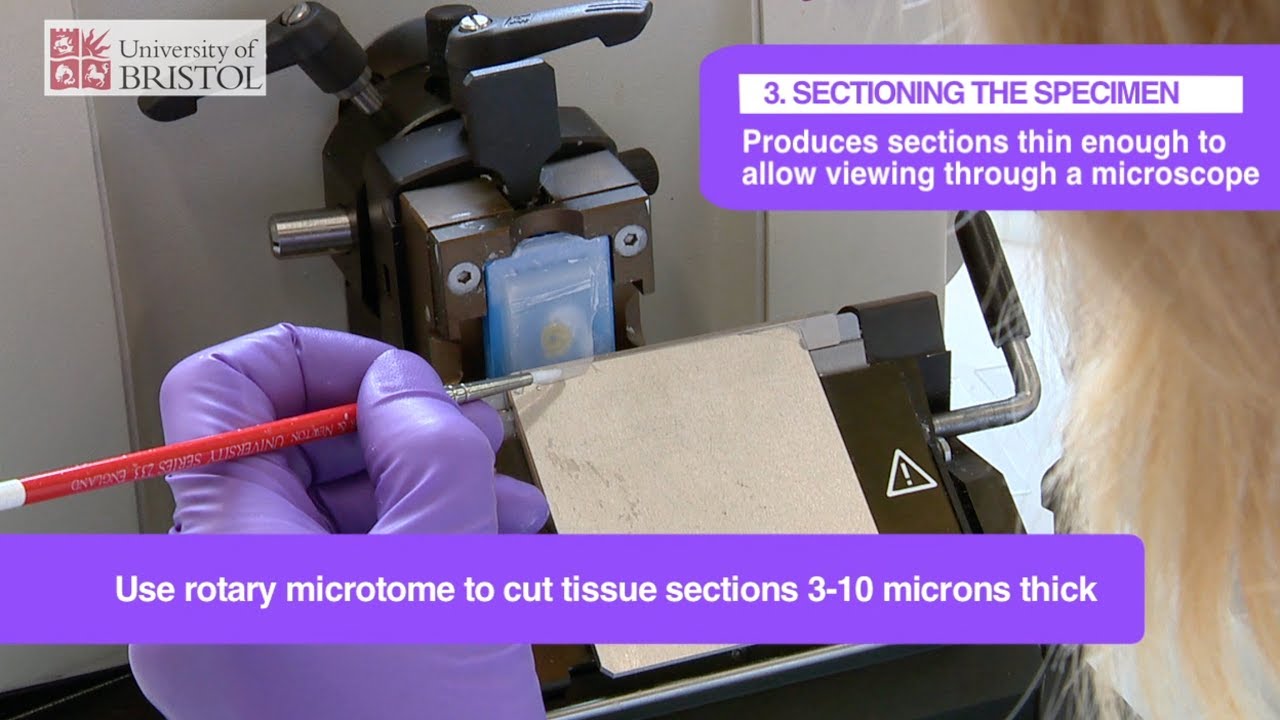
STEP 3 — Sectioning (Microtomy)
- Rotary microtome: most common; paraffin block trimmed and sectioned at 3–5 µm
- Ribbon: consecutive sections form a ribbon of sections floating on water bath (40–45°C) — tissue expands and sections are mounted on labelled glass slides
- Drying: slides dried at 60°C oven overnight or 1 hour to adhere sections
- Frozen sections (cryostat): tissue snap-frozen in liquid nitrogen or dry ice; sectioned at −20 to −30°C; 5–8 µm; results in 15–20 minutes; used for intraoperative diagnosis, enzyme/immunofluorescence studies; lower quality
STEP 4 — Deparaffinisation and Rehydration
Before aqueous staining: xylene (removes wax) → descending alcohols → distilled water.
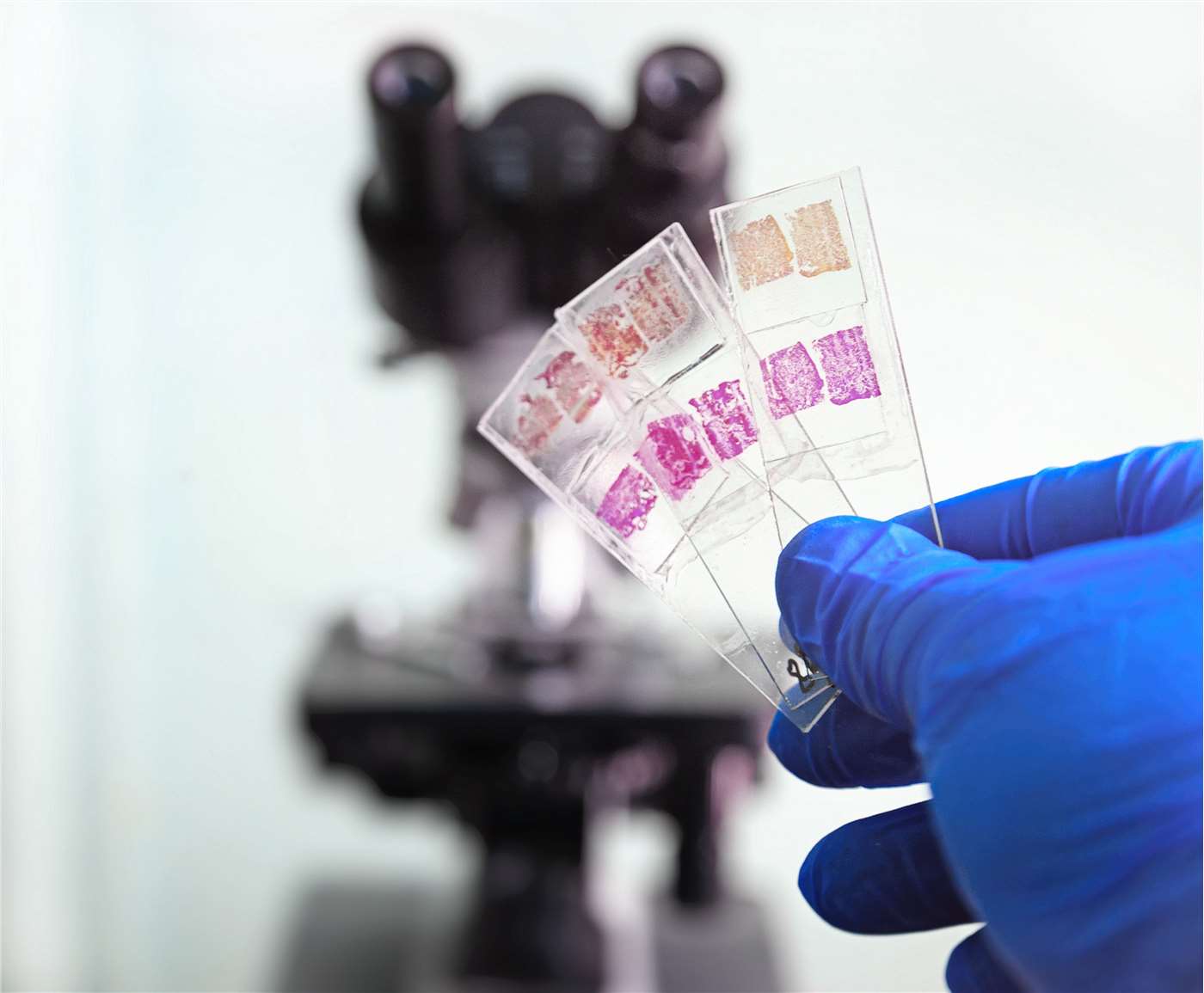
STEP 5 — Staining
A. Haematoxylin & Eosin (H&E) — Routine Stain
The gold standard stain for histological diagnosis. Two components:
- Haematoxylin: natural dye derived from logwood; oxidised to haematein; acts as a basic dye forming complexes with alum mordant; stains acidic structures blue/purple (nuclei — DNA, RNA; cartilage matrix; calcium; mucin)
- Eosin: synthetic acidic dye; stains basic structures pink (cytoplasm, collagen, muscle fibres, red blood cells, most connective tissue)
- Process: haematoxylin → rinse → differentiation in acid alcohol → bluing in tap water → eosin → dehydrate → clear → mount (DPX)
B. Special Stains — Summary Table
The table below lists commonly used special stains, their targets, colour results, and typical applications.
| Stain | Targets | Colour Result | Common Applications |
|---|---|---|---|
| H&E | Nuclei, cytoplasm, ECM | Nuclei blue/purple; cytoplasm/ECM pink | Routine histology; general morphology |
| PAS | Glycogen, mucin, basement membranes, fungi | Magenta | Liver (glycogen), kidney (basement membranes), fungal infections |
| Van Gieson | Collagen vs. muscle | Collagen red; muscle/yellow | Fibrosis, scar tissue, blood vessels |
| Masson's Trichrome | Collagen, muscle, nuclei | Collagen blue/green; muscle red/pink; nuclei black | Liver fibrosis, cardiac pathology, muscle biopsy |
| Silver (Reticulin) | Reticular fibres (Type III collagen) | Black | Lymphoid organs, liver (sinusoids), bone marrow |
| Orcein / Weigert's | Elastic fibres | Brown/black | Arteries (elastin), lung, skin |
| Ziehl–Neelsen | Acid-fast bacteria | Red | Tuberculosis, leprosy |
| Gram | Bacteria (Gram +/-) | Gram+ purple; Gram- pink | Bacterial infections in tissues |
| Oil Red O / Sudan IV | Lipids | Red/orange | Fat stains (requires frozen sections) |
| Prussian Blue (Perls) | Ferric iron (haemosiderin) | Blue | Iron overload (hemochromatosis), bone marrow |
| Alcian Blue | Acid mucopolysaccharides (GAGs) | Blue | Cartilage, goblet cell mucin, some tumours |
C. Immunohistochemistry (IHC)
Antibodies conjugated to enzymes (peroxidase, alkaline phosphatase) or fluorochromes detect specific antigens in tissue sections. Widely used for tumour typing (cytokeratins for carcinoma, CD markers for lymphoma), receptor status (ER, PR, HER2), and infection (HPV, CMV).
STEP 6 — Mounting and Coverslipping
DPX (distrene plasticiser xylene) or equivalent resinous mountant applied; coverslip placed; dried. Slides permanently preserved for archive review.
Quality Control in Slide Preparation
- Inadequate fixation → nuclear detail lost; cytoplasm poorly defined
- Over-dehydration → brittle sections; tearing and folding
- Thick sections (>8 µm) → nuclear overlap; difficult interpretation
- Air bubbles under coverslip → artefact
- Contamination → false-positive results (especially IHC)
Video Lesson
Slide Preparation & Staining — Histology Video Lesson
Click to play video lesson
References
4 sources- 1
Bancroft JD, Layton C. Bancroft's Theory and Practice of Histological Techniques (8th ed.). Elsevier; 2019.
- 2
Ross MH, Pawlina W. Histology: A Text and Atlas (8th ed.). Wolters Kluwer; 2020.
- 3
Young B, O'Dowd G, Woodford P. Wheater's Functional Histology (6th ed.). Churchill Livingstone/Elsevier; 2014.
- 4
Suvarna SK, Layton C, Bancroft JD. Theory and Practice of Histological Techniques (7th ed.). Churchill Livingstone/Elsevier; 2013.
Disclaimer: These notes are for educational purposes only and compiled from standard histology textbooks. Clinical interpretation of slides requires a qualified histologist or pathologist.
Ready to test yourself?
Apply what you've learned in the Histology Spotting Test