PharmaceuticalTechnology
Advanced drug delivery science — from barriers to drug delivery and conventional dosage form principles through novel targeting strategies, NDDS characterisation techniques, pharmacokinetic/pharmacological aspects, and commercial applications of advanced drug delivery systems.
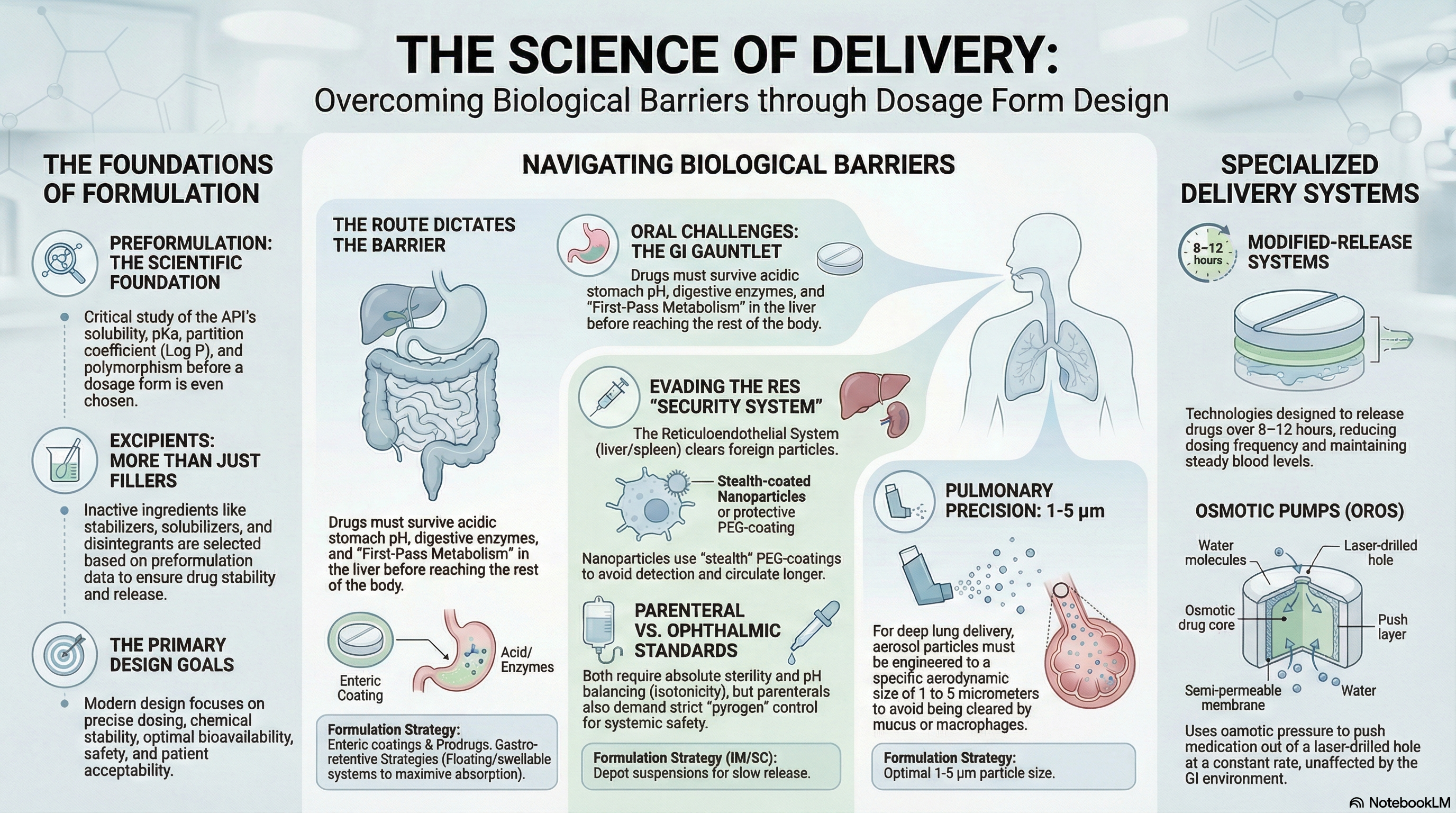
Barriers & Conventional Formulations
Physiological and biochemical barriers to drug delivery (GI, blood-brain, skin, ocular, nasal), principles of conventional pharmaceutical formulations, dosage form design, biopharmaceutics classification system (BCS), and ADME considerations in formulation development.
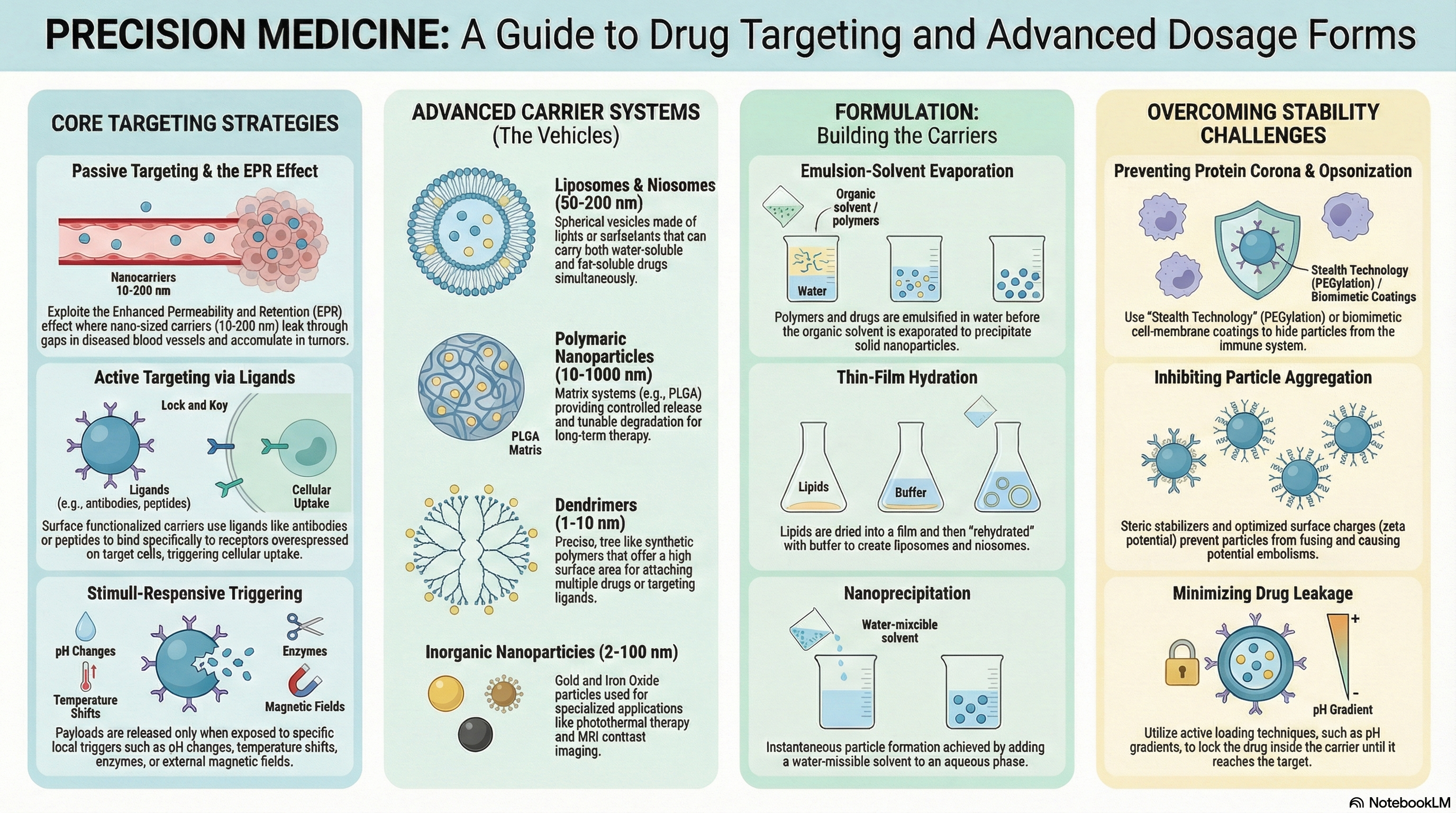
Targeting & Advanced Dosage Forms
Drug targeting concepts (passive, active, physical targeting), liposomes, nanoparticles, microspheres, niosomes, transfersomes, solid lipid nanoparticles (SLNs), dendrimers, carbon nanotubes, transdermal drug delivery, implants, and pulsatile drug delivery systems.
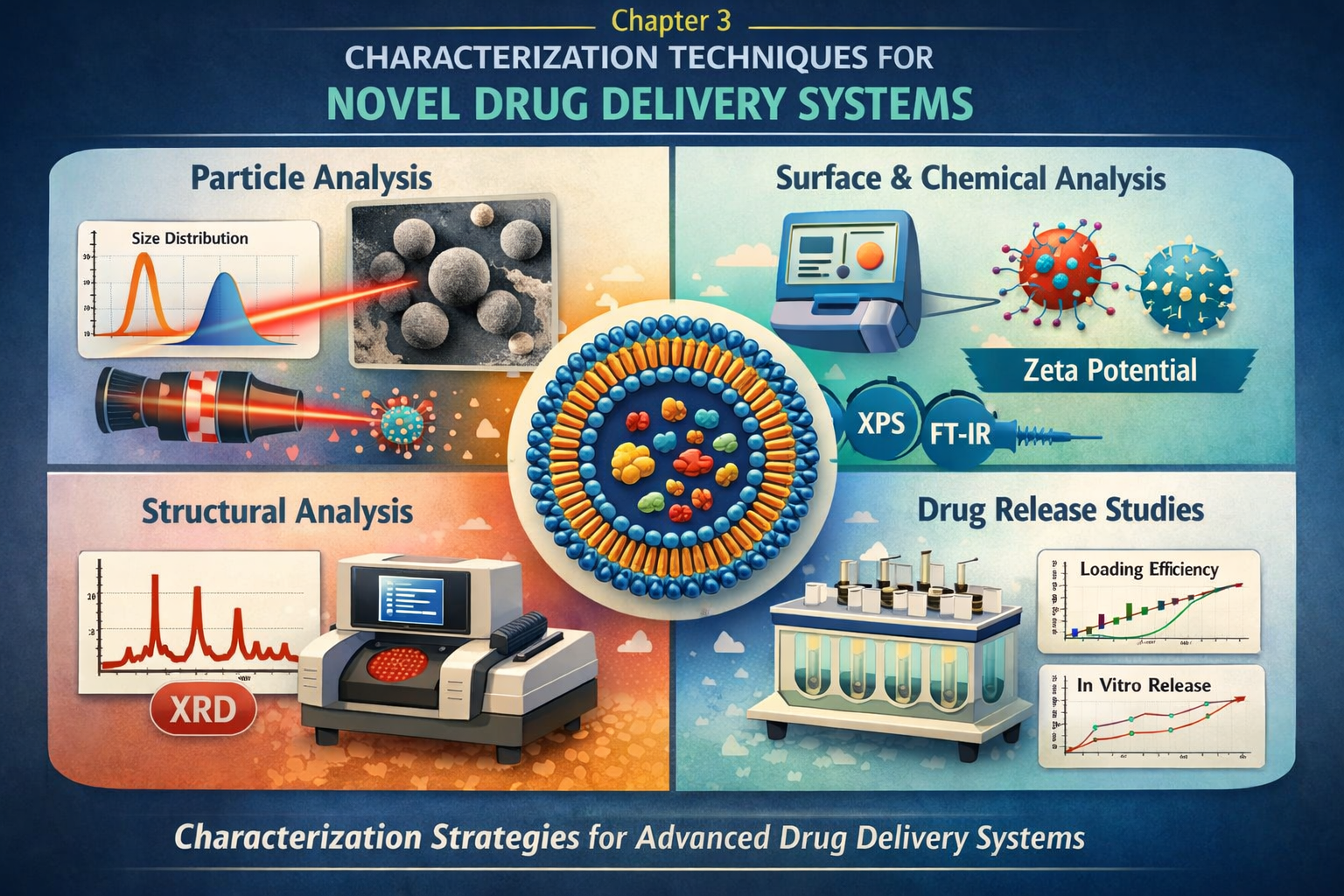
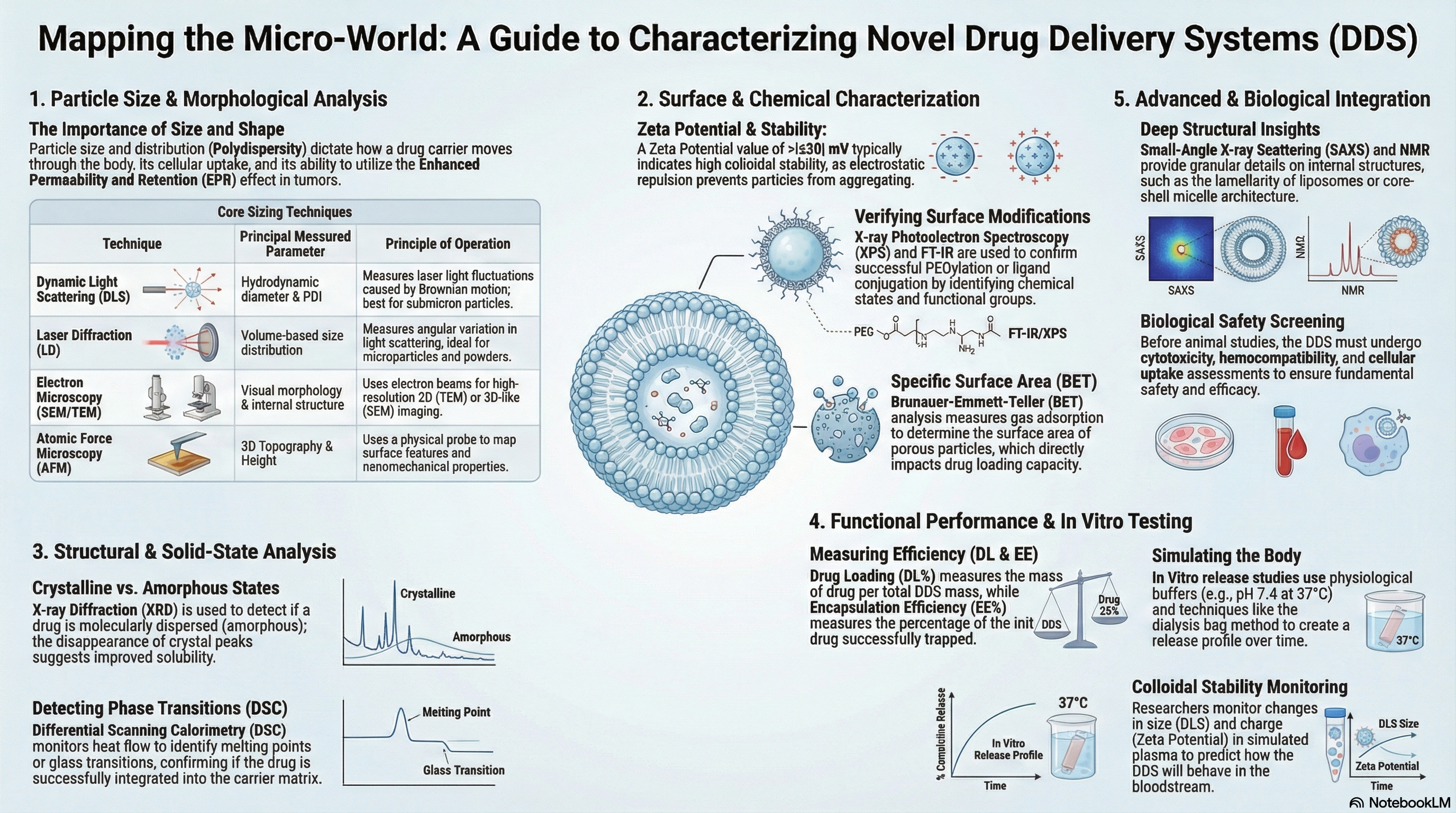
Characterisation of NDDS
Physicochemical characterisation of NDDS — particle size analysis (DLS, SEM, TEM), zeta potential, entrapment efficiency, drug release studies (dialysis, Franz diffusion cell), in vitro-in vivo correlation (IVIVC), stability testing, and in vitro cytotoxicity assays.
PK/PD Aspects of NDDS
Pharmacokinetics of novel drug delivery systems — compartmental models, non-compartmental analysis, bioavailability and bioequivalence, modified release kinetics (zero-order, first-order, Higuchi, Korsmeyer-Peppas), pharmacodynamic considerations, and toxicokinetics of nanoparticles.
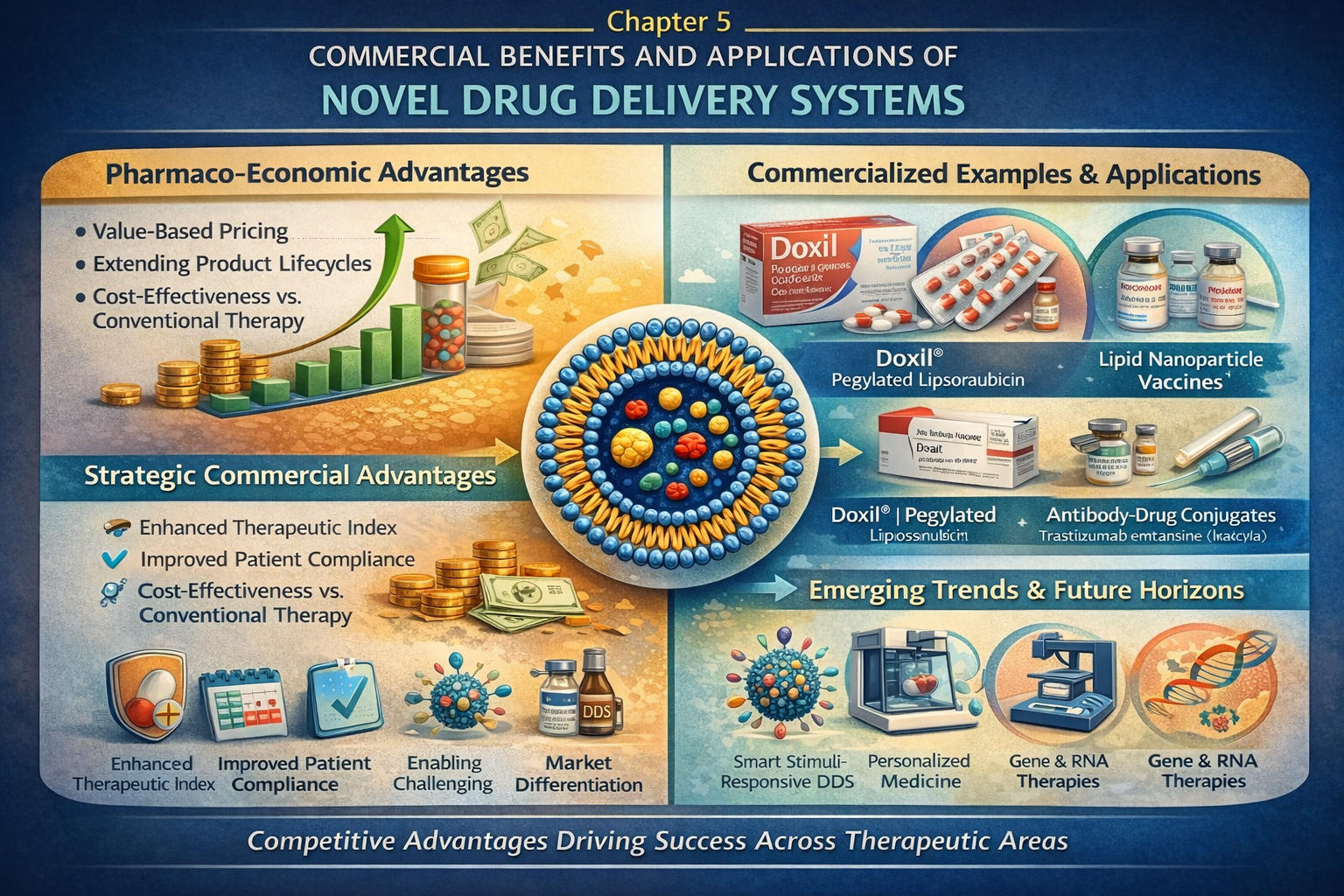
Commercial Benefits of NDDS
Market landscape of novel drug delivery systems, patent strategies, life-cycle management, commercial products (Doxil, Abraxane, Neulasta, Ozempic depot), regulatory pathway for NDDS approval (FDA, EMA guidelines), cost-benefit analysis, and future trends in the NDDS market.
Start with Chapter 1 — Barriers & Conventional Formulations