PharmaceuticalOrganic Chemistry
The chemical language of drug design — from atomic structure and bonding through functional groups and reaction mechanisms to stereochemistry and its critical role in pharmacological activity.
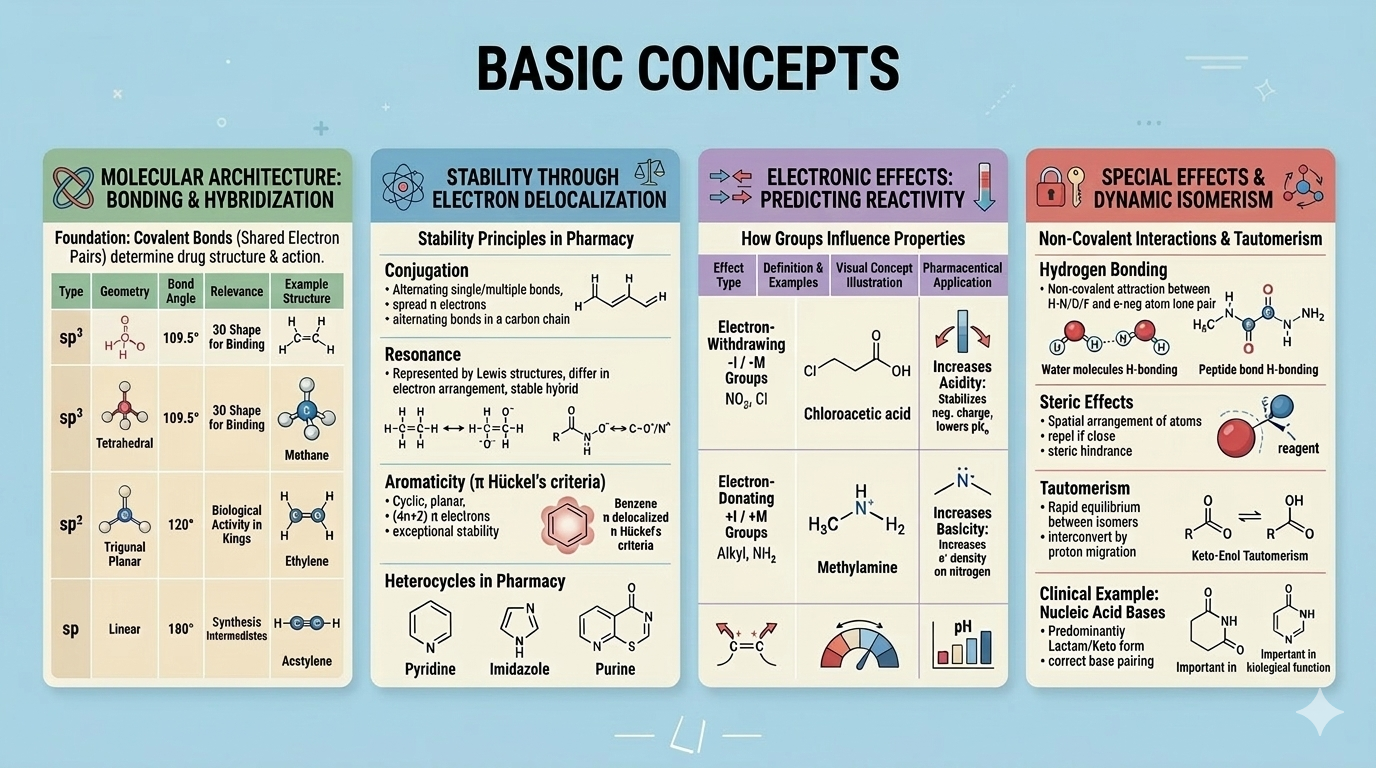
Basic Concepts
Atomic orbitals, hybridisation (sp, sp², sp³), covalent bonding, resonance, inductive and mesomeric effects, electronegativity, acids and bases in organic chemistry, and IUPAC nomenclature.
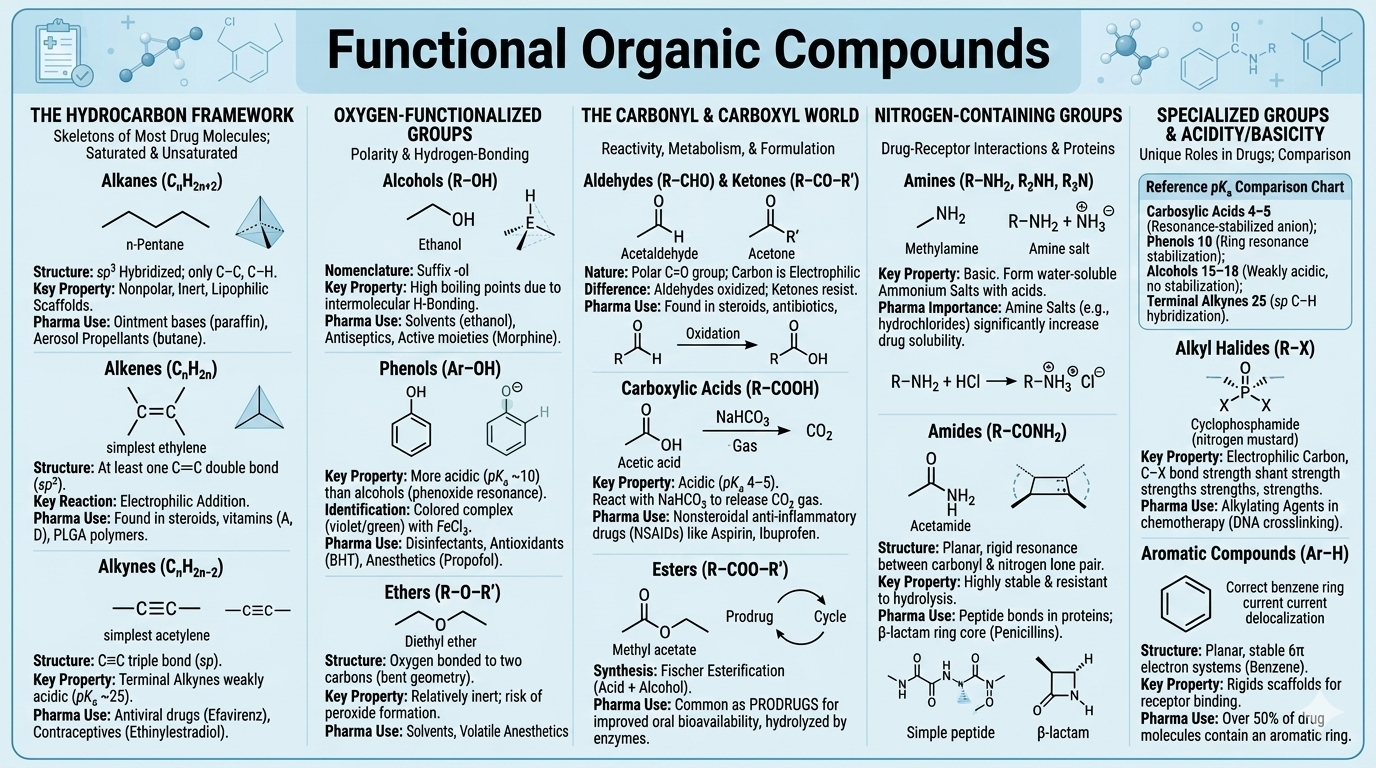
Functional Organic Compounds
Hydrocarbons, alkyl halides, alcohols, phenols, ethers, aldehydes, ketones, carboxylic acids, esters, amines, and amides — structures, properties, and pharmaceutical relevance.
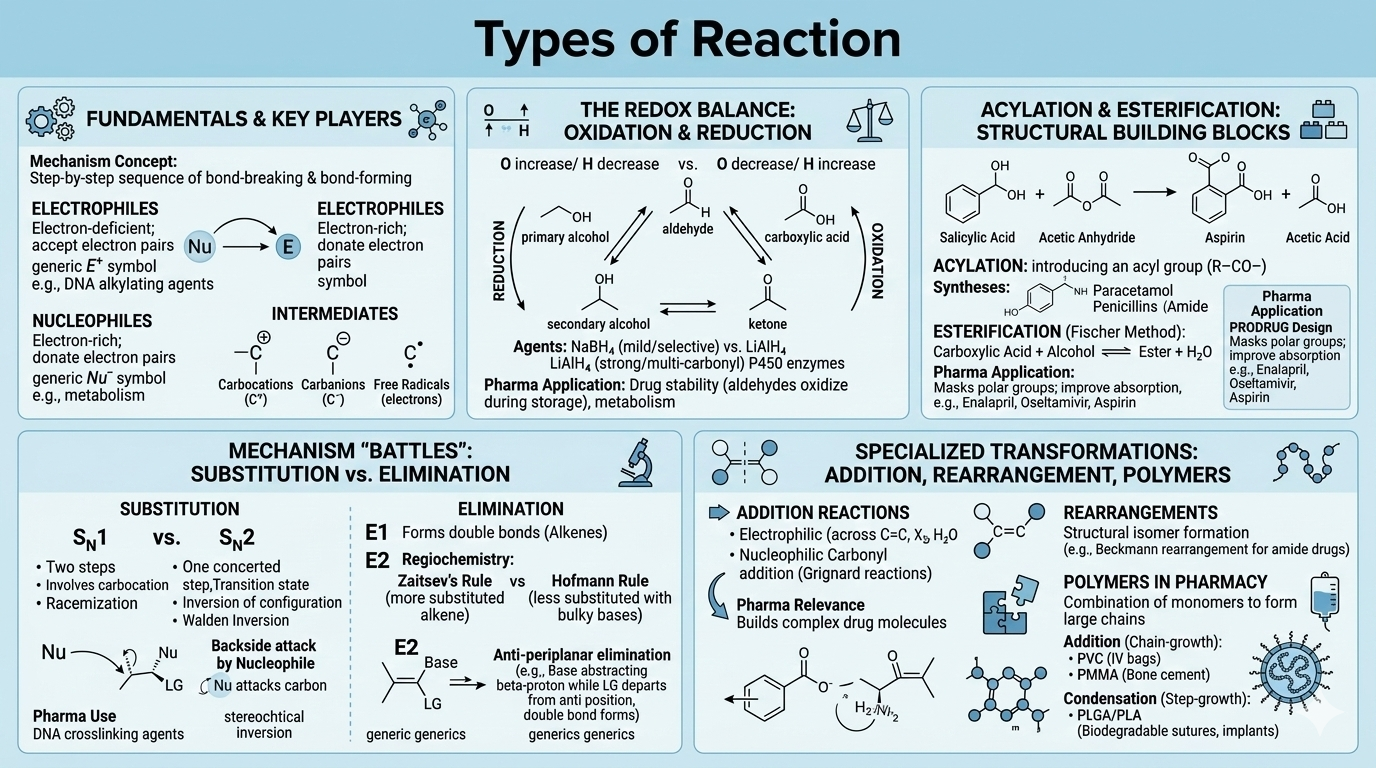
Types of Reactions
Substitution (SN1, SN2), elimination (E1, E2), addition, condensation, and oxidation-reduction reactions. Reaction mechanisms, intermediates, energy diagrams, and catalysis.
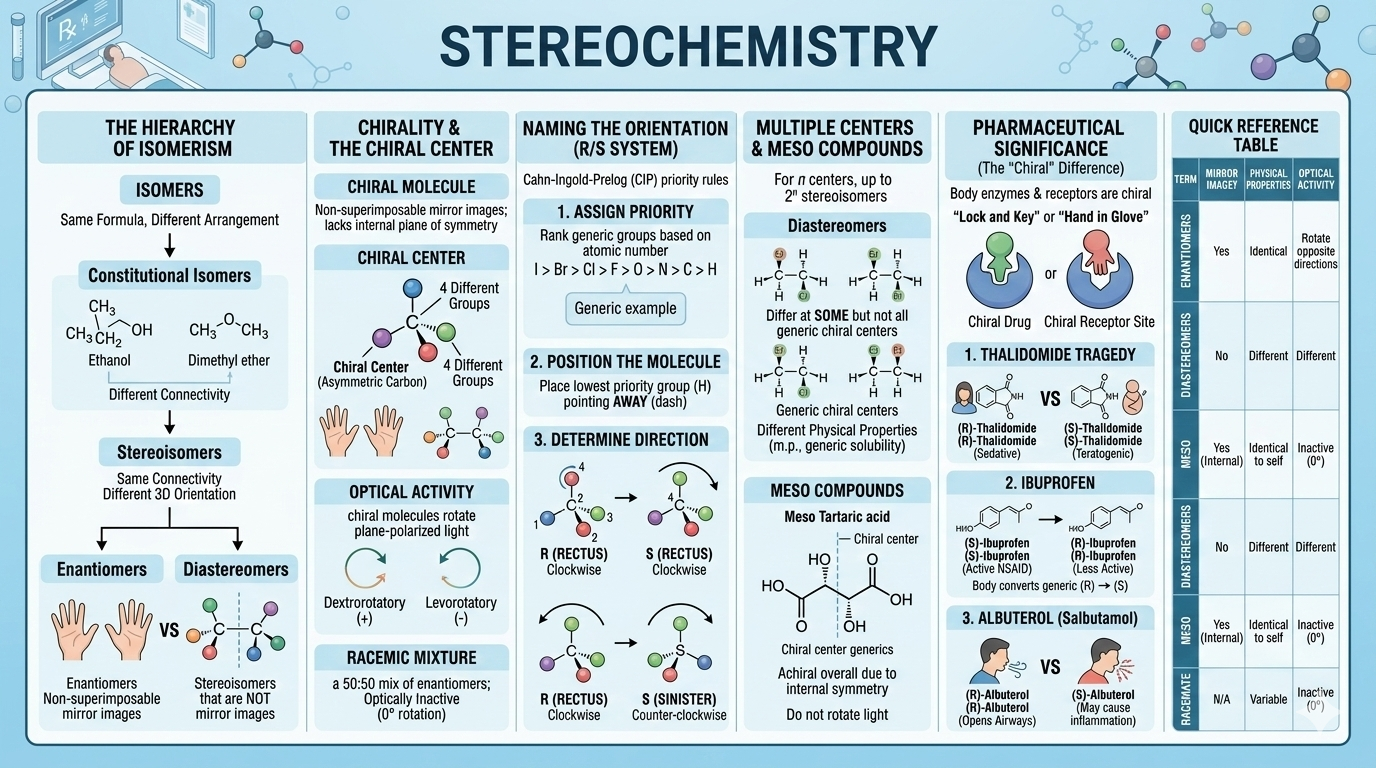
Stereochemistry
Chirality, enantiomers, diastereomers, R/S and E/Z configuration, optical activity, racemic mixtures, meso compounds, and the pharmacological significance of stereoisomerism in drug action.
Start with Unit 1 — Basic Concepts and work your way up.